2.5 Atomic and Nuclear Physics - Introduction
|
By the end of this unit you should be able to:
In addition to the above criteria, we may also explore aspects of:
Lecture Notes for the whole unit
|
| ||||||
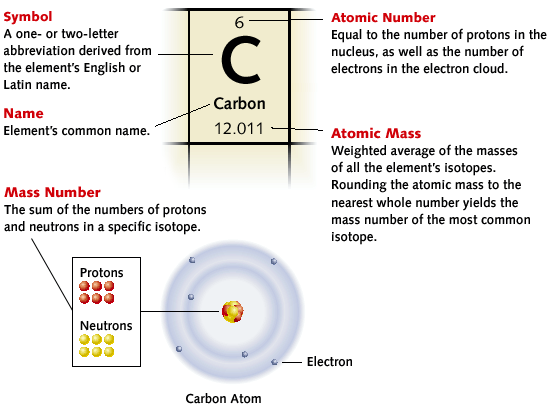
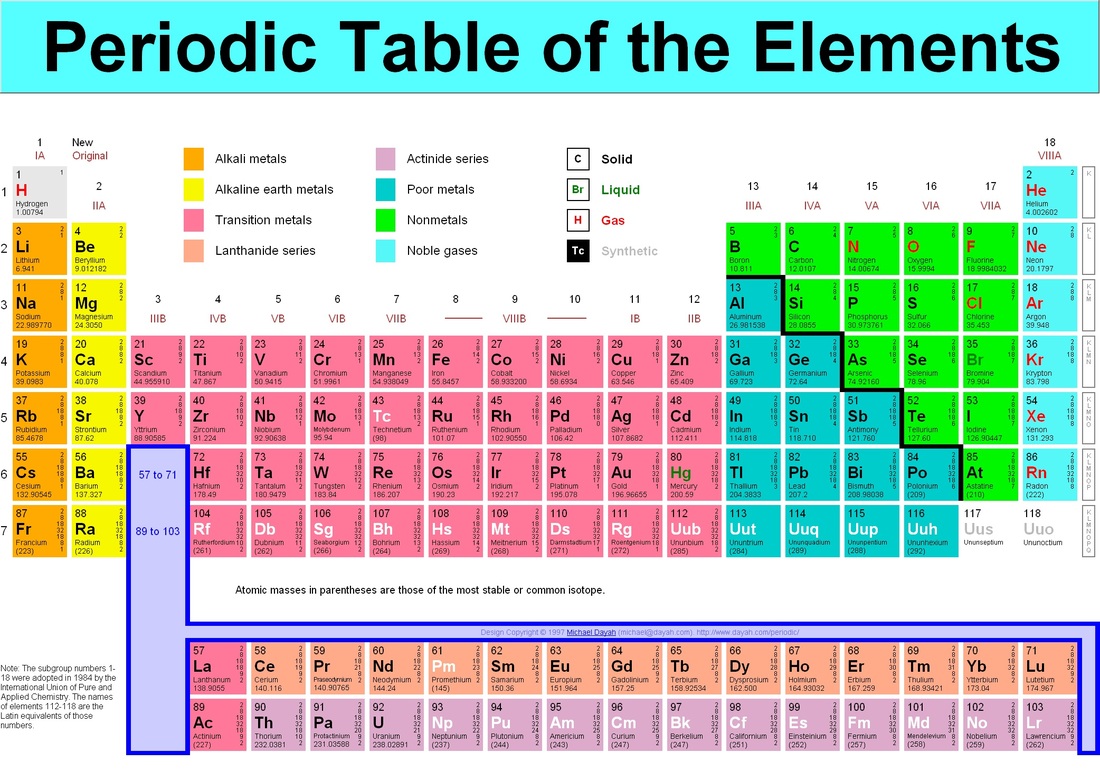
2.5.1 Atoms, Isotopes, Periodic Table
|
Lesson Overview:
Homework: Pgs 9-12
|
|
2.5.2 Historical Models of the Atom
|
Lesson Overview:
There have been a number of historical models that describe the structure of the atom. Our focus will be on John Dalton, J.J. Thomson, and Ernest Rutherford (see next section)
HOMEWORK: Pages 48-49
|
|
2.5.3 Rutherford's Experiment
|
Lesson Overview:
Sir Ernest Rutherford discovered the positively charged nucleus during his famous "gold foil" experiment. Key points from his results include:
|
|
2.5.4 Radioactivity
|
Lesson Overview:
There are three types of radiation: Alpha, Beta, Gamma. They are all ejected from the nucleus of an unstable atom and result in an atom with different properties.
HOMEWORK: Text 56, 59-63
|
|
2.5.5 Nuclear equations And Conservation of Mass
|
Lesson overview:
Two main principles are concerned with nuclear reactions 1) Conservation of mass: The total mass of the left and right side of any equations will be the same. 2) Conservation of charge: The total charge on the left and right side of any equations will be the same. These principles can be used to determine the reactant, product, or type of decay, when two of these are known. Activity:
Text page 64-65 |
|
2.5.6 Half-Life
|
Lesson Overview:
Half life is defined as the amount of time it takes for half of a sample to decay. This could be measured by mass or with a Geiger counter in clicks per minute. Activity: Text page 66-69 |
|
2.5.7 Nuclear Reactions: Fission and Fusion
|
Lesson overview:
Fission is when a nucleus breaks into two or more parts creating new, smaller elements and releasing lots of energy. Conservation of mass and charge apply when balancing nuclear fission equations. Fusion is the fuzing of nuclei (usually small nuclei such as hydrogen) to make large elements. This is the reaction the provides the energy in most stars. Activity 1: Fission Text page 72-73 Activity 2: Fusion and Nuclear Power Text page 74 |
|
2.5.8 Mass and Energy Equivalance
|
Lesson Overview:
When energy is released during a nuclear reaction, a small amount of total mass is also lost. This can be calculated using E=mc^2. This shows that matter can be converted to energy and energy can be converted to matter. The constant c is the speed of light (3.00x10^8 m/s) so there is an enormous amount of energy contained in even a very small amount of mass. Activity: Text page 70-71 |
|
2.5.9 Uses of Radioactivity
|
Textbook Page: 75-79
|
|
2.5.-- Extra: Quantum Mechanics and Particle Physics
|
This sect ion is above and beyond what we need to know for this unit but it may shed some light on some of the how and why we observe particles behaving the way they do.
|
|
|
Exam Review
Textbook End of Chapter Review pages 80-88
Answers to past exam questions below. Also find lots of notes and study material at No Brain Too Small.
| atoms__atomic_structure_q_and_a.pdf | |
| File Size: | 197 kb |
| File Type: | |
| atoms__types_of_radioactivity__answers.pdf | |
| File Size: | 159 kb |
| File Type: | |
| atoms__half_life_q_and_a.pdf | |
| File Size: | 347 kb |
| File Type: | |